|
Dick White Frits |
Zinc Silicate Crystalline Glaze Pottery A chronicle of my recent progress and a way for me to keep it straight in my head! |
![]() Thread:
Frit comparison | Forum:
Crystalline
Glaze | Posted by: Dick White | Date: May 9th, 2009 08:24 PM
Thread:
Frit comparison | Forum:
Crystalline
Glaze | Posted by: Dick White | Date: May 9th, 2009 08:24 PM
Ok Kat, here it is.
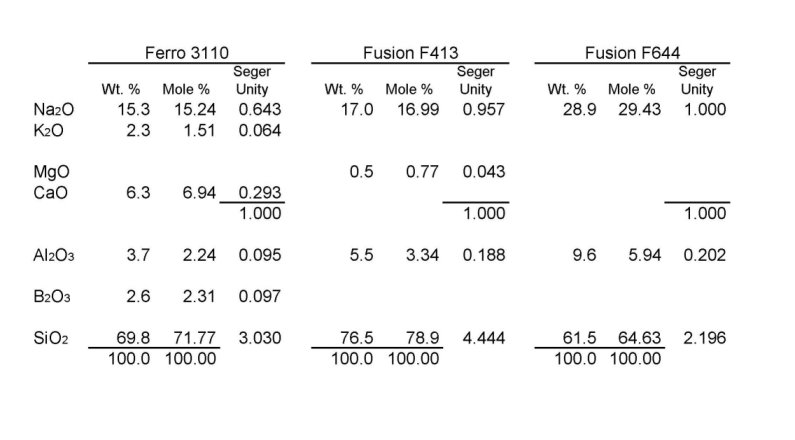
For each of the frits you asked about, I've provided
the percentage by weight, the percentage by moles, and the Seger Unity analysis
as if the glaze contained only that frit. The percentage by weight is exactly
what it would seem. Of 100g of 3110, you would find 15.3g of sodium oxide, 2.3g
of potasium oxide, 6.3g of calcium oxide, etc. The molar percentages convert the
weight distribution to the proportion of molecules of each compound based on the
atomic mass of each compound. The unity analysis is where it gets interesting.
For this, simply sum the molar percentages of the fluxes and then determine a
scaling factor to adjust the sum to 1. For the 3110, the sum of the Na, K, and
Ca molar percentages is 23.69. To convert that to 1, you would multiply by
0.042219037. Now that you have the adjusting factor to convert total fluxes to
1, multiply each of the individual molar percentages by that number. Now you
have a set of numbers that tell you that for every 1000 molecules of flux, 643
of them will have Na, 64 of them will have K, and 293 of them will have Ca
(i.e., roughly 2/3 Na and 1/3 Ca with a touch of K thrown in). And further, for
each 1000 molecules of flux, you will find 95 molecules with Al, 97 molecules
with B, and 3030 molecules with Si.
Shifting over to the Fusion F413, the flux is 96% Na and 4% Mg. Comparing this
to the 3110, Na is a stronger flux than Ca, so substituting the same amount of
3110 vs 413 in your glaze will result in more fluxing with the 413. It doesn't
stop there, however. Looking at the other components, for each 1000 molecules of
flux, the 413 has about double the number of Al atoms but only 1.5 times the
number of Si atoms. This important for us, because we need to keep the Al to a
minimum so the glaze will be fluid enough for the crystals to grow. At the same
time, we need an excess of Si (compared to "normal" glazes) so there will enough
of that to join with the Zn to form the zinc orthosilicate crystals we want. So,
if we start substituting 413 for 3110 in a known good recipe, we are adding Al
at a faster rate than Si, so we'll also need to increase the raw Si in the glaze
recipe to compensate. The fluxing action will also change.
Looking at the 644, the flux is entirely Na. While this may be a bit like the
413 which was 95% Na, the Al and Si numbers are moving in the other direction.
Compared to 3110, the 644 has double the Al, about like the 413, but only 2/3
the Si. Thus you would need to really dump in the extra raw Si if you were to
substitute an equal amount of 644 for 3110.
All this is complicated by the Zn requirements in our crystalline glazes. We
need to keep the ratio of Zn atoms to Si atoms within a fairly narrow range for
the crystal to ground balance to come out the way we want. That means you will
need to adjust the Zn in your recipe too as you change frits.
Finally, the wild card in all of this is the boron, which we touched on in one
of the other threads. Notice that 3110 is the only one that has any B. If your
recipe is 413 or 644 based, you will need an alternate source of B if you want
any of that. Gerstley borate or Ferro 3134 are good sources for that without
altering the rest of the chemical balance too much.
So there it is. Have fun with it
(Edited by Dick White)
Phil Hamling
376 County Route 1
Warwick, NY, USA 10990